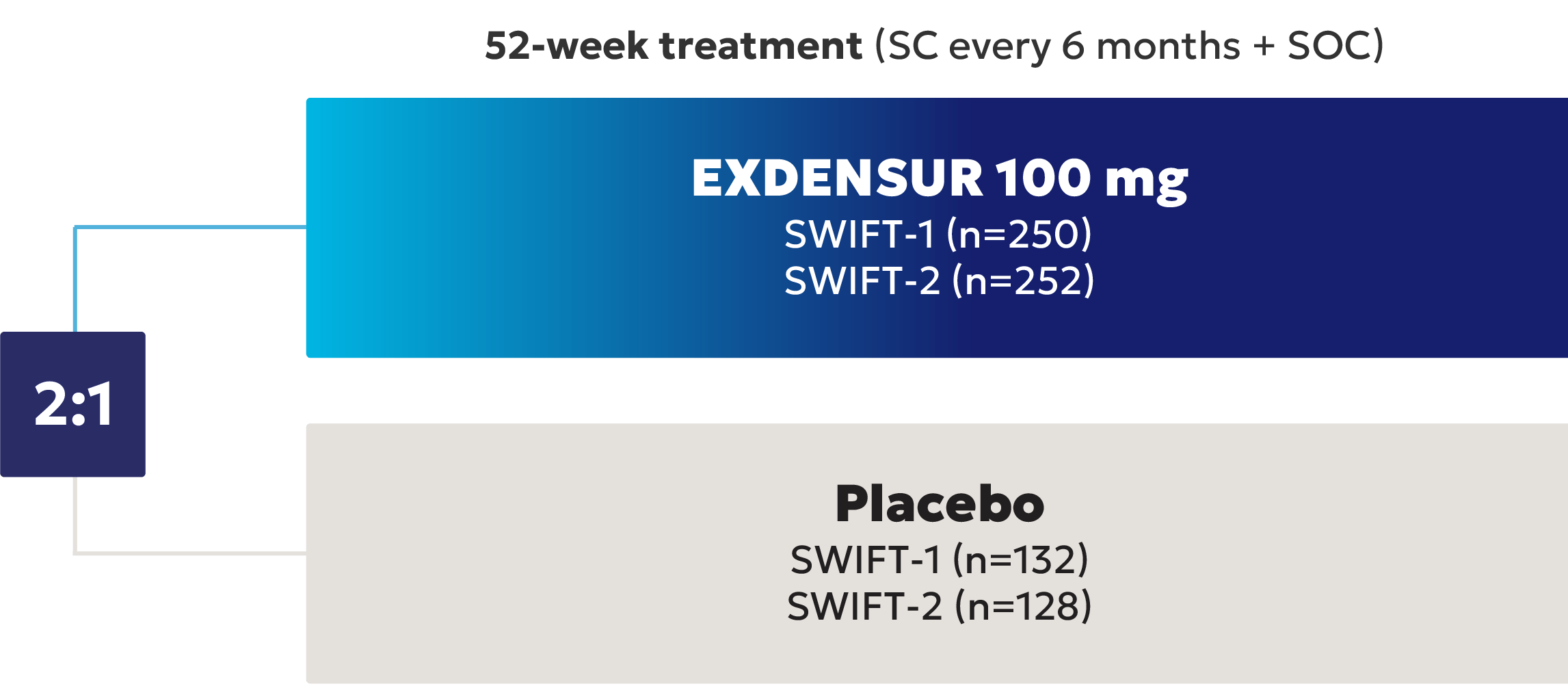Indication & Safety Info
Indication
Important Safety Information
Indication
EXDENSUR is indicated for the add-on maintenance treatment of severe asthma, characterized by an eosinophilic phenotype, in adult and pediatric patients aged 12 years and older. EXDENSUR is not indicated for the relief of acute bronchospasm or status asthmaticus.
Important Safety Information
WARNINGS AND PRECAUTIONS
Hypersensitivity Reactions
Hypersensitivity reactions, including anaphylaxis, can occur following administration of EXDENSUR. If a hypersensitivity reaction occurs, discontinue EXDENSUR and initiate appropriate therapy.
Acute Asthma Symptoms or Deteriorating Disease
EXDENSUR should not be used to treat acute asthma symptoms or acute exacerbations.
Risk Associated with Abrupt Reduction of Corticosteroid Dosage
Upon initiation of EXDENSUR therapy, do not abruptly discontinue systemic or inhaled corticosteroids. Reductions in corticosteroid dose, if appropriate, should be gradual and under the supervision of a healthcare provider. Reduction in corticosteroid dose may be associated with systemic withdrawal symptoms and/or unmask conditions previously suppressed by systemic corticosteroid therapy.
Parasitic (Helminth) Infection
Patients with pre-existing helminth infections should be treated for their infection prior to initiation of EXDENSUR therapy. If patients become infected while receiving EXDENSUR and do not respond to anti-helminth treatment, discontinue EXDENSUR until the infection resolves.
ADVERSE REACTIONS
In patients receiving EXDENSUR, the most common adverse reactions (≥4%) were upper respiratory tract infection, allergic rhinitis, influenza, arthralgia, and pharyngitis. Injection site reactions have also occurred.
USE IN SPECIFIC POPULATIONS
The data in pregnant women are insufficient to identify a drug-associated risk of major birth defects, miscarriage, or other adverse maternal or fetal outcomes. Transport of endogenous IgG antibodies and monoclonal antibodies, such as depemokimab-ulaa, across the placenta increases as pregnancy progresses and peaks during the third trimester.
EXDENSUR can cross the placenta during pregnancy and the presence of the YTE modification may prolong and increase exposure to the infant exposed in utero. The impact of transmission to the fetus should be considered. Pregnant women exposed to EXDENSUR, or their healthcare providers, should report EXDENSUR exposure by calling 1-888-825-5249.
Please see full Prescribing Information for EXDENSUR.